Author: R&D Team, CUIGUAI Flavoring
Published by: Guangdong Unique Flavor Co., Ltd.
Last Updated: Feb 25, 2026
Every food scientist and R&D manager knows the sinking feeling: the “gold standard” benchtop prototype—the one that wowed the marketing team and aced early sensory panels—tastes completely different after its first run through the pilot plant. The vibrant top notes are muted, an unexpected caramelized note has appeared, or the overall profile feels “flattened.”
This is the central challenge of industrial food production: bridging the gap between culinary artistry and manufacturing reality.
As professional manufacturers of food and beverage flavorings, we live at this intersection. We understand that flavor is not a static ingredient; it is a dynamic, volatile entity that reacts violently to the stresses of manufacturing. The journey from raw ingredient to packaged product is fraught with chemical perils, most notably the “Big Three” processing parameters: Heat, Pressure, and pH.
Mastering flavor stability requires more than just buying a good flavor; it requires a deep technical understanding of the mechanisms at play during processing. This article provides a technical deep dive into how these forces alter flavor profiles and offers insights into how we, as your flavor partners, formulate to survive the gauntlet of industrial production.

Target Flavor vs. Industrial Processing Reality
To understand how processing impacts flavor, we must first establish what we are trying to protect. Flavor is a complex neurophysiological response combining gustation (taste: sweet, salty, sour, bitter, umami) on the tongue and olfaction (smell) in the nasal cavity.
The vast majority of what we perceive as “flavor nuance” comes from olfaction, driven by Volatile Organic Compounds (VOCs). These are small, light molecules—esters, aldehydes, ketones, terpenes, and pyrazines—that easily evaporate at room temperature, traveling to our olfactory receptors.
The fundamental problem with processing is that these VOCs are inherently unstable. They are sensitive to energy input (heat and pressure) and chemical environments (pH). When subjected to processing, three things generally happen to these vital compounds:
Understanding these pathways is the first step toward controlling them. Let us examine the primary drivers.
Heat is the most common, and most destructive, force applied in food processing. Used for pasteurization, sterilization (UHT/canning), baking, and dehydration, thermal energy provides the activation energy required for countless chemical reactions that alter flavor.
Not all thermal effects are negative. Heat is responsible for the desirable “cooked” flavors we associate with many foods.
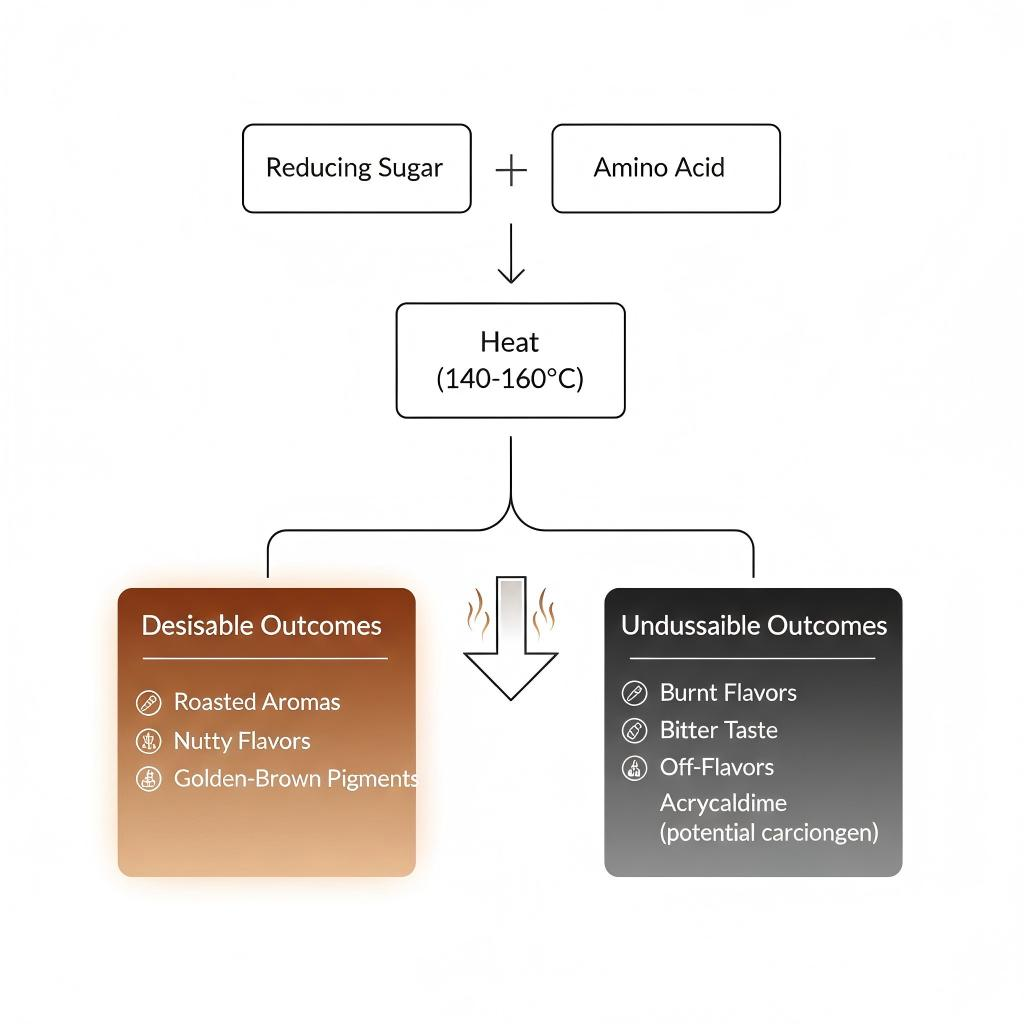
The most significant of these is the Maillard reaction, a complex series of non-enzymatic browning reactions between reducing sugars and amino acids (proteins). This reaction, occurring readily above 140°C (284°F), is responsible for the savory, roasted, nutty, and meaty notes in coffee, chocolate, baked goods, and seared meats. It generates thousands of flavor compounds, including pyrazines, pyrroles, and furans.
Similarly, caramelization occurs when sugars are heated (pyrolysis) in the absence of amino acids, resulting in sweet, nutty, and sometimes bitter flavor compounds distinct from the Maillard reaction.
However, in products designed to taste “fresh”—like a strawberry yogurt, a citrus beverage, or a delicate herbal sauce—these reactions are detrimental. A fresh strawberry flavor profile relies on delicate esters (like ethyl butyrate) and aldehydes. Under high heat (e.g., UHT pasteurization for a shelf-stable beverage), two things happen: the fresh esters flash off or hydrolyze, and the natural sugars and proteins in the fruit juice base initiate Maillard browning.
The result is a product that tastes like “cooked jam” rather than fresh fruit. The profile shifts from bright and fruity to heavy, sweet, and slightly caramelized.
Heat also accelerates lipid oxidation, particularly in products containing fats and oils. This is a free-radical chain reaction that degrades unsaturated fatty acids. While sometimes desirable in small amounts for fried food profiles, unchecked lipid oxidation leads to rancidity.
In processed meats, reheating can trigger “warmed-over flavor” (WOF), characterized by cardboard-like or metallic off-notes. This is primarily due to the oxidation of membrane phospholipids, accelerated by heat and the release of iron from myoglobin during cooking.
The most direct impact of heat is simply increasing the kinetic energy of VOCs, causing them to escape the food matrix. The “top notes” of a flavor—the immediate, impactful scents that define freshness—are usually the lowest molecular weight and lowest boiling point compounds. They are the first casualties of thermal processing.
According to sources like the American Chemical Society, understanding the thermodynamics of these volatile compounds is crucial, as their loss changes the fundamental ratio of the flavor profile, often leaving only the heavier, slower-releasing “base notes” behind.
Industry Insight: When formulating for high-heat applications (like baked goods or hard candies), we rarely use the same flavor “recipe” as for a cold application. We must “over-dose” the sacrificial top notes, knowing many will be lost, and rely heavily on precursors that generate flavor during the heating process, rather than adding finished volatiles that will just evaporate.
Maillard Reaction Chemical Pathway Diagram
While often used in conjunction with heat, pressure itself is a powerful thermodynamic parameter that uniquely influences flavor chemistry. In modern food manufacturing, pressure is generally applied in two very different ways: thermal retort (canning) and High-Pressure Processing (HPP).
Traditional canning or retort processing uses steam under pressure to achieve temperatures well above boiling (typically 121°C / 250°F) to sterilize low-acid foods.
In this scenario, pressure acts primarily to allow water to reach superheated temperatures without boiling away. The flavor impacts here are essentially extreme versions of thermal processing described above. The combination of intense heat and moisture under pressure accelerates hydrolysis reactions and pushes Maillard reactions very quickly. This is why canned vegetables or soups have a distinct “retort flavor”—a generalized cooked, slightly sulfurous profile where individual ingredient nuances are melded into a uniform background.
High-Pressure Processing (HPP), or pascalization, is a different beast entirely. It is a non-thermal pasteurization technique where food, already sealed in its final package, is subjected to enormous isostatic pressure (often 300–600 MPa) transmitted by water.
HPP is celebrated for its ability to inactivate spoilage microorganisms and vegetative bacteria without the use of high heat. Because covalent bonds (the strong bonds holding flavor molecules together) are generally unaffected by pressure at these levels, HPP excels at preserving small volatile flavor compounds.
A fresh cold-pressed juice processed via HPP will retain significantly more of its “raw” flavor profile, vibrant color, and nutritional content compared to a thermally pasteurized counterpart.
However, HPP is not entirely flavor-neutral. While it spares covalent bonds, pressure does disrupt weak non-covalent bonds, such as hydrogen bonds and hydrophobic interactions. This can lead to changes in the food matrix structure:
The FDA notes that while HPP is effective for pathogen control and preserving sensory attributes better than thermal treatments, its effects on the food matrix’s physical structure must be considered during product development.
Industry Insight: Designing flavors for HPP requires a different mindset. We don’t need to compensate for thermal loss, but we must ensure the flavor system is stable in the specific aqueous environment of the product and won’t interact negatively with pressure-modified proteins or residual enzyme activity.

Digital pH Measurement of an Acidic Beverage
While heat and pressure are external forces applied to the product, pH is an intrinsic property of the food matrix itself. The acidity or alkalinity of a product acts as a master switch, determining which chemical reactions can occur and how we perceive the resulting flavors.
pH impacts flavor in three distinct ways: chemical stability, enzymatic control, and sensory perception.
Many critical flavor compounds are pH-sensitive. Esters, responsible for nearly all fruity flavors, are prone to acid hydrolysis.
In a low-pH environment (like a carbonated soft drink or a sour candy with a pH below 3.0), esters will slowly break back down into their constituent alcohols and acids in the presence of water. A bright “ripe banana” flavor (isoamyl acetate) will, over time in an acidic beverage, degrade into isoamyl alcohol (fusel oil smell) and acetic acid (vinegar smell).
This degradation is highly time and temperature-dependent. A beverage might taste perfect at bottling, but after six months on a warm warehouse shelf, the acid hydrolysis will have completely shifted the flavor profile toward a “chemical” or “fermented” off-note.
Acidity also dictates the color stability of anthocyanins (the red/purple pigments in berries), which are often inextricably linked to consumer perception of flavor. A shift in pH can turn a vibrant red strawberry drink into a dull purple one, negatively influencing the perceived flavor before the consumer even tastes it.
Enzymes are the biological catalysts of flavor change, and every enzyme has an optimal pH range. By controlling pH, processors can inhibit undesirable enzymatic reactions.
For example, lipoxygenase is an enzyme that catalyzes the oxidation of fatty acids, leading to “beany” or “grassy” off-flavors in soy or oat milks. Adjusting the pH away from this enzyme’s optimum range during processing is a key strategy for mitigating these off-notes.
Finally, pH has a direct impact on how flavor molecules interact with taste receptors.
Acidity (sourness) itself is a primary taste, but it also modifies the perception of other tastes. Acid suppresses sweetness; therefore, a higher acid product requires more sugar to achieve the same perceived sweetness level.
Furthermore, the ionization state of many flavor molecules changes with pH. Some compounds are only volatile (and therefore aromatic) in their non-ionized state. If the pH of the food shifts them into an ionized state, they become less volatile and the aroma intensity drops. Research in sensory science frequently highlights how manipulating the acid/base balance of a food matrix can radically alter the release kinetics of volatile aroma compounds during consumption.
The real challenge in industrial processing is that these factors rarely occur in isolation. They work in synergy, often compounding their destructive effects.
A flavor that is stable under heat might fall apart if the pH is lowered. A flavor that works in a neutral pH UHT beverage might turn bitter in an acidic hot-fill application.
Given these hostile environments, how do we, as flavor manufacturers, ensure that the mango flavor in your finished, pasteurized, shelf-stable beverage still tastes like a fresh mango?
We don’t just sell liquids in bottles; we engineer flavor delivery systems adapted to your specific processing parameters. Here are the core strategies we employ:
This is perhaps the most powerful tool in our arsenal. Encapsulation involves trapping volatile flavor cores within a protective “wall” material (typically modified starches, gums, or proteins). This wall acts as a barrier against heat, oxygen, and reactive components in the food matrix.
According to industry reviews, such as those found in Food Technology Magazine, advanced microencapsulation is essential not just for stability, but for controlling the timing of flavor release—ensuring the flavor bursts during consumption, not during the retort process.
For high-heat applications like baking or savory snack production, adding highly volatile finished aroma compounds is inefficient. Instead, we utilize flavor precursors.
We can formulate a flavor system containing specific reducing sugars, amino acids, and fats that are relatively inert at room temperature. When your product hits the oven, these precursors react in situ via the Maillard reaction and lipid degradation to generate the desired flavor profile right at the moment of consumption. We are essentially using your processing oven as the final step in flavor manufacturing.
We rarely offer “off-the-shelf” solutions for complex processing. We need to know your matrix.
Knowing these parameters allows us to select aroma chemicals with higher boiling points or greater resistance to acid hydrolysis. We might substitute a delicate natural strawberry ester with a more robust, naturally-derived analogue that survives your specific pasteurization curve.
Sometimes the simplest solution is strategic application. If we know a process strips 30% of top notes, we may formulate the flavor with top notes 30% higher than the target profile.
Alternatively, we work with process engineers to change when the flavor is added. Injecting flavor aseptically after the heating step, rather than before, is an expensive infrastructure upgrade for a manufacturer, but it often solves the most difficult flavor stability problems.

Food Scientist Scaling Flavor Technology
The journey from a delicious concept to a commercially viable, processed food product is treacherous for flavor molecules. Heat transforms, pressure disrupts, and pH acts as a volatile gatekeeper.
If you are finding that your scaled-up product lacks the vibrancy of your R&D prototype, the issue likely isn’t the quality of the flavor—it’s the incompatibility of that flavor with your processing reality.
Don’t treat flavor as an afterthought or a simple commodity ingredient. It is a complex chemical system that needs to be engineered for your specific manufacturing environment.
At CUIGUAI Flavor, we don’t just create great flavors; we ensure they survive your process. Our team of flavor chemists and food technologists is ready to analyze your specific heat, pressure, and pH parameters to develop a robust, customized flavor solution.
Are you struggling with flavor loss during scale-up? Contact our technical team today for a consultation or request a processing-stable sample tailored to your application.
| Contact Channel | Details |
| 🌐 Website: | www.cuiguai.cn |
| 📧 Email: | info@cuiguai.com |
| ☎ Phone: | +86 0769 8838 0789 |
| 📱 WhatsApp: | +86 189 2926 7983 |
| 📍 Factory Address | Room 701, Building 3, No. 16, Binzhong South Road, Daojiao Town, Dongguan City, Guangdong Province, China |
Selected Natural Citations for Reference (to be hyperlinked in final publication):
Copyright © 2025 Guangdong Unique Flavor Co., Ltd. All Rights Reserved. Return and Exchange Policy